Biography
I am an accomplished epidemiologist, who specializes in the principled use and development of advanced causal inference methods for answering pharmacepideiological research questions in numerous therapeutic areas. I currently work at Amgen, a large biotech company, where I support the bone health franchise’s generation of real-world evidence on the benefit:risk profile of bone products and related regulatory engagements. Prior to joining Amgen, I was an epidemiologist at Target RWE, where I generated real-world evidence studies in the dermatology and oncology therapeutic areas. I received an MPH and a PhD in Epidemiology from Columbia University Mailman School of Public Health where I had formal training in epidemiological methods with a primary focus on causal inference methods.
Download my CV.
- Linking claims and clinical data
- Causal inference methods in pharmacepidemiology
- Osteoporosis and bone pharmacoepidemiology
PhD in Epidemiology, 2023
Columbia University
MPH in Epidemiology, 2016
Columbia University
BA in Business communications, 2010
Richmond University
Skills
Experience
Major responsibilities include:
- Regulatory Real-World Evidence (RWE) & PMR Leadership
- Led design and authorship of FDA-required studies, including a post-marketing requirement final report, NCO study protocols/SAPs, and algorithms validating 3-point MACE and CV mortality using Medicare–REGARDS linked data
- Developed and led the clean-room gating framework with quantitative assessments of potential unmeasured confounding to support upcoming comparative safety work
- Conducted multiple ad-hoc analyses integrating claims and biometric EHR data through multiple imputation and transportability methods, demonstrating negligible unmeasured confounding due to biometric covariates in upcoming claims-only safety study
- Ran simulation studies evaluating bias from nondifferential outcome misclassification and applied validation-based adjustments to recover true effects
- Authored several regulatory briefing documents and RTQ responses, ensuring methodological clarity and alignment with FDA expectations
- Additional Regulatory, Methodological, and Cross-Functional Contributions
- Co-led protocol development for the Effectiveness NCO study, aligning core design elements with the Safety NCO study and adding two new negative control outcomes
- Supported global regulatory needs through regulator-mandated safety analyses across U.S. and Japan datasets, identifying cross-regional methodological efficiencies
- Contributed to Japan PMO/MOP work through study feasibility assessments and creation of an R Shiny app for automated power/sample size estimation across multiple testing frameworks
- Authored key sections of two briefing documents for Japanese biopharma regulator, clarifying misclassification bias and differential attrition concerns in regulator-conducted studies
- Coauthored two manuscripts on postoperative outcomes among women with osteoporosis and supported broader publication strategy with bone and publication teams
- Evaluated PCORNet network performance to optimize site selection for future claims–EHR linked analyses
- Served as a reviewer for Amgen’s internal protocol governance body
Responsibilities included:
- Designing pharmacoepidemiological hepatology, dermatology, and oncology studies with claims and electronic health record data, including studies that integrate both types of data
- Implementing advanced causal inference methods to surmount common problems in pharmacoepidemiological analyses
- Clone censor weight approach to address immortal time bias due to the start of follow-up and treatment initiation not coinciding
- Inverse probability of censoring weight approach for informative right censoring
- Prevalent new user design for non-contemporaneous marketing in comparative effectiveness studies
- Writing proposals and developing presentations to help Target RWE win new contracts and continue existing ones
- Completed doctoral epidemiology training and dissertation on nicotine vaping’s unintended consequences
- Performed teaching assistant duties for numerous epidemiology and statistics courses
Apps
Featured Project
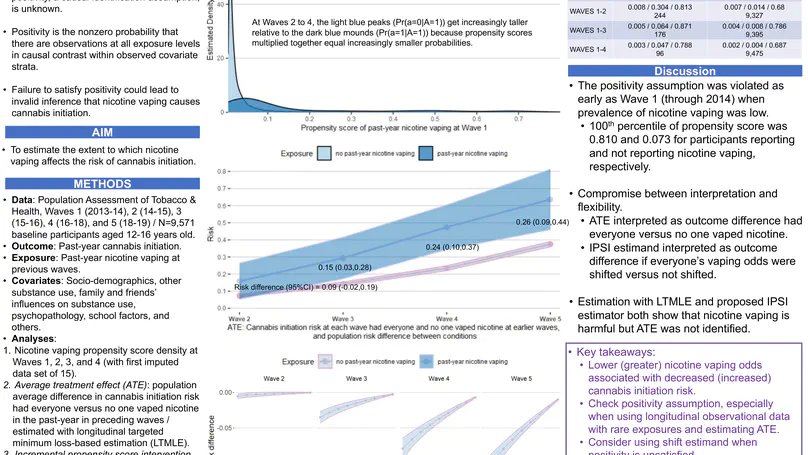
In a longitudinal study of nicotine vaping’s influence on cannabis initiation, we found that the positivity causal identification assumption was violated. Regardless, we still estimated the average treatment effect (ATE) and an analogue estimand that does not require the positivity assumption. The analogue, the incremental propensity score intervention (IPSI), was the difference in cannabis initiation risks had everyone’s odds of nicotine vaping been decreased up to 90% compared with observed nicotine vaping odds. Interpetations of results from the IPSI and the ATE were consistent with each other. That is, lower odds of nicotine vaping decreases cannabis initiation risks (IPSI), and nicotine vaping increases cannabis initiation risk (ATE). Researchers should consider using shift estimands like IPSI when the positivity assumption is violated.